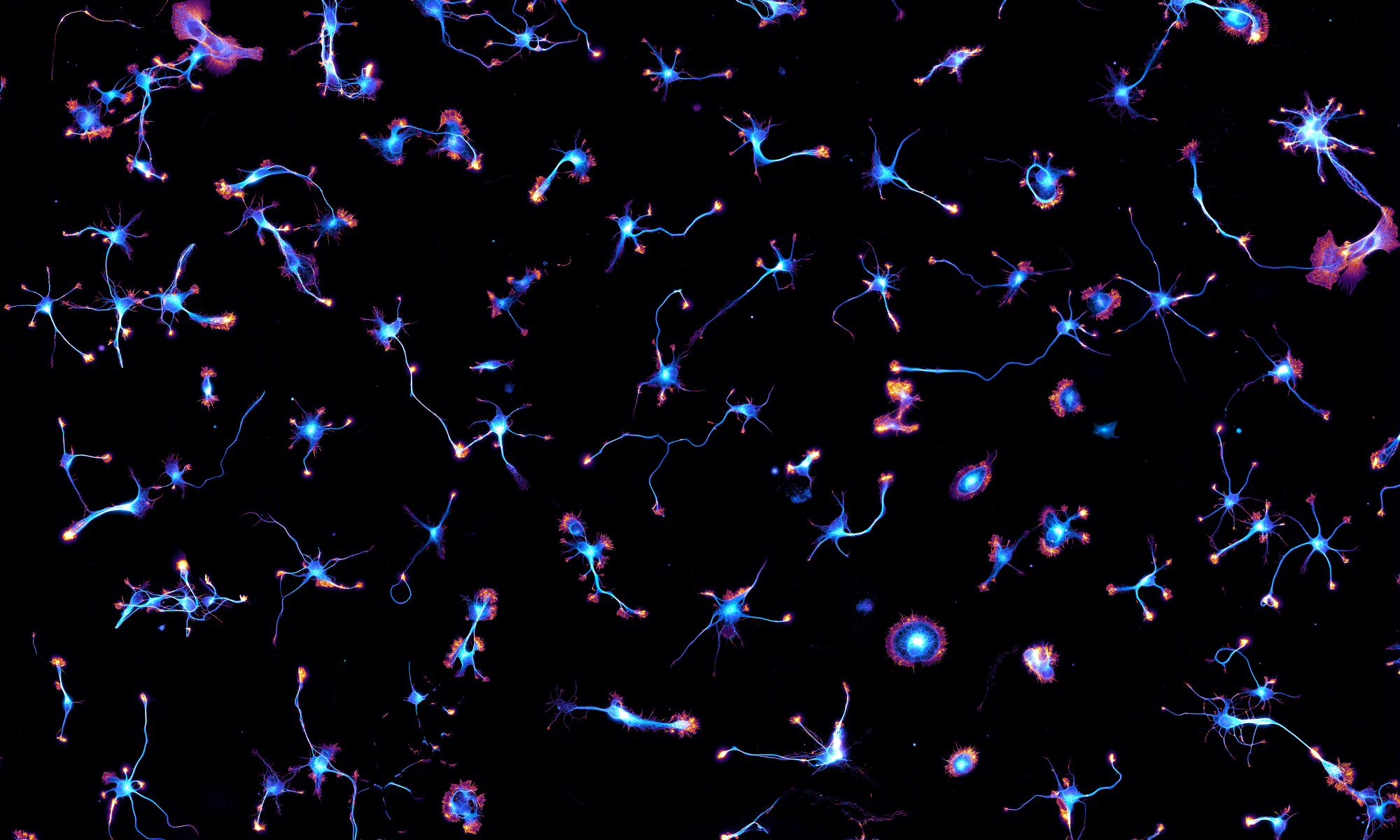
For the last two weeks, we got to play with Nikon Instruments latest super-resolution spinning disk microscope that incorporates a Yokogawa SoRa head. It was installed in the INP NeuroCellular Imaging Service (NCIS) imaging facility.

We could make 3D-stacks and live-cell imaging movies of cells and neurons benefiting form the ~120 nm lateral resolution. See the how this compares to diffraction-limited imaging with this example from our samples: